Chapter 7 Nuclear Properties
7.1 Discovery of the nucleus
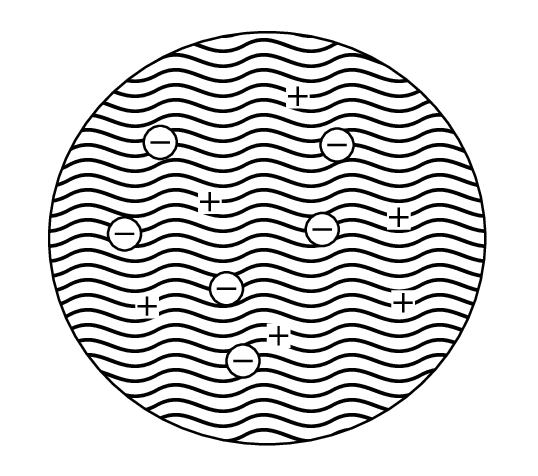
Early experiments showed that electrons and unidentified positive particles could be emitted from atoms. It was therefore reasonable to assume that atoms consisted of + and - charges and Thomson (who discovered the electron) suggested that atoms were spheres of positive charge of the order of m in radius (roughly the atomic radius) in which electrons were embedded.
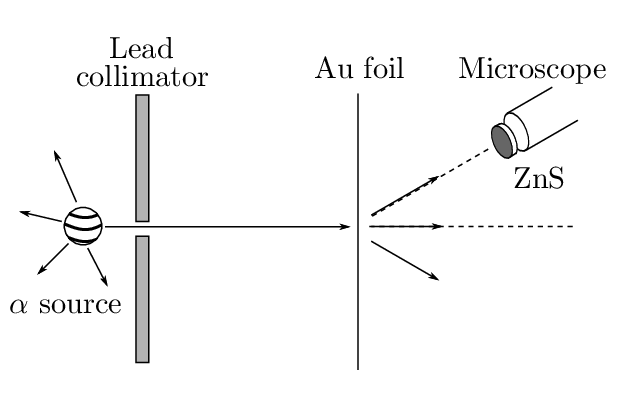
In 1909 Geiger and Marsden tested the Thomson model by scattering a beam of particles from a gold film ( particles were used as they were available from radioactive decay of unstable nuclei). The ‘plum pudding’ model implies small angle scattering. (The probability of an particle being scattered by more than in the model is 1 in ). This is because many scattering events (each with a small scattering angle) are required to produce a large scattering angle. However, about 1 particle in underwent scattering through – an enormous discrepancy. Rutherford is quoted as saying ‘it was as if you fired a 15-inch shell at a sheet of tissue paper and it came back at you!’
The small scattering in the Thomson model results from the small electric fields in the atom. This led Rutherford to propose in 1911 a model with a small positively charged core, the nucleus, surrounded by atomic electrons. Since the electrons are much lighter than an atom, most of the mass is concentrated in the nucleus. The cross-section for scattering can be calculated under the following assumptions
-
1.
A Coulomb interaction between the particle and nucleus (assuming both are point particles).
-
2.
Scattering is elastic.
-
3.
The target is thin enough to consider only single scattering.
Elastic Coulomb scattering is called Rutherford scattering. Calculation of the differential cross-section is more lengthy than that of hard sphere scattering considered in lecture 6, and is found to be
| (7.1) |
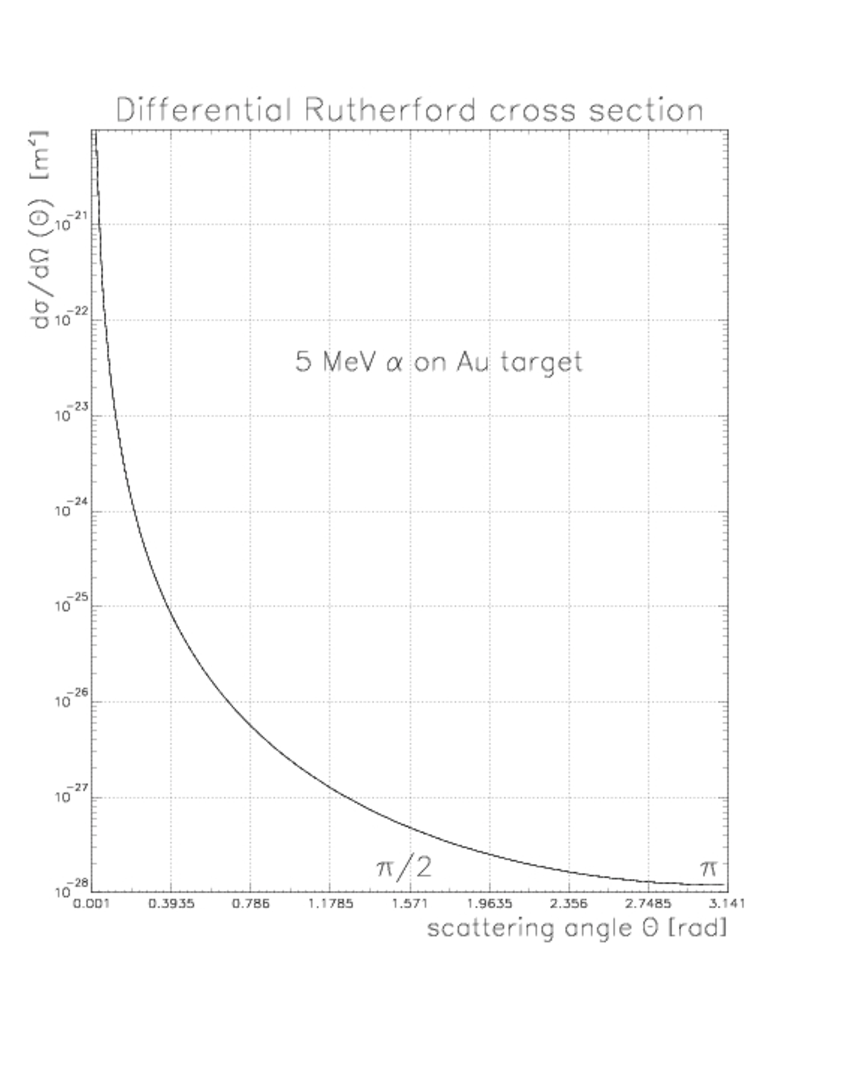
where for an particle. Here and are the mass and velocity of the particle. Notice the scattering is strongly peaked in the forward direction (). Also notice the differential cross-section is proportional to , target thickness and . The differential cross-section is plotted in Fig. 7.3.
These dependencies were all confirmed by Geiger and Marsden. The discovery of the nucleus can be regarded as the birth of Nuclear Physics.
7.2 Parity and magnetic moment
We know protons have the following basic properties
-
Mass: 1.672621637 kg
-
Electric charge: +1e
-
Spin: 1/2
and neutrons
-
Mass: 1.67492729 kg
-
Electric charge: 0
-
Spin: 1/2
There are further numbers to describe particles. We will discuss 2 of these – parity and magnetic moment.
7.2.1 Parity
Definition 7.2.1 (Parity Operation (P)).
The parity operation reflects all spatial coordinates through the origin. In Cartesian coordinates:
| (7.2) | |||
and in spherical polar coordinates
| (7.3) | |||
In general, a physical system is left unchanged by a parity operation and none of its observable properties or physical laws change, so
| (7.4) |
This leaves two possibilities:
| (7.5) | |||
The ‘+’ sign implies a symmetric wavefunction and corresponds to ‘even’ parity. The ‘-’ sign implies an antisymmetric wave function and corresponds to ‘odd’ parity.
Parity is fixed for particular particles which are said to have ‘intrinsic parity’. Particles with half integer spins have opposite parity to their anti-particles whereas particles with integer spin have the same parity as their anti-particles. Thus the parity is even for electrons and nucleons but odd for positrons and anti-nucleons.
The parity of a system of 2 or more particles is given by the algebraic product of the intrinsic parities and an angular momentum term. The parity contribution due to orbital angular momentum is even for even and odd for odd. We can write this term as . The parity of a 2 particle system is therefore
| (7.6) |
where and are the intrinsic parities of each particle.
7.2.2 Parity conservation
If we were to find a system with we would conclude the system is not invariant under parity. No evidence has been found that the strong nuclear force or electromagnetic interactions violate parity.
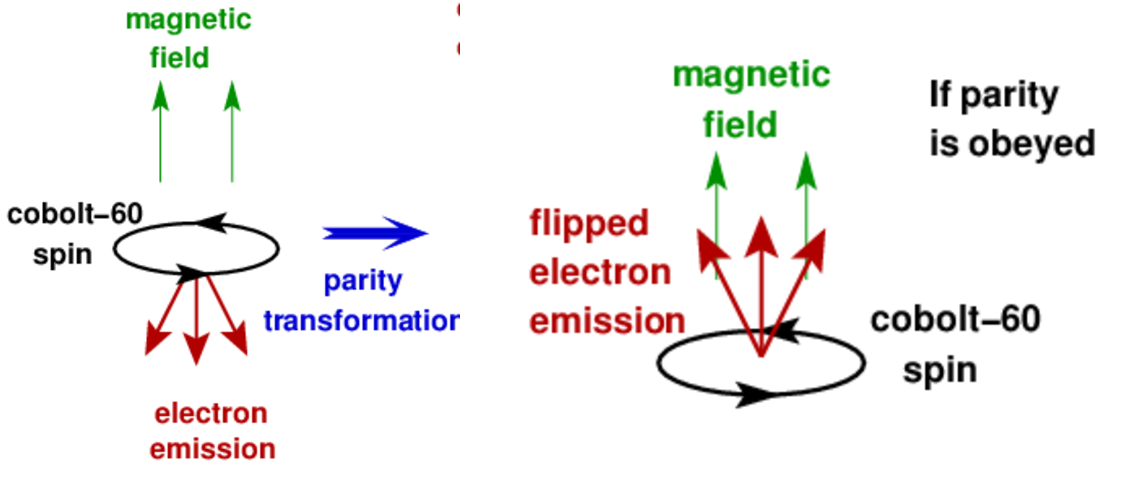
The weak nuclear force (which involves beta decay), however, does violate parity. This was discovered in 1957 by Wu, who was studying beta decay of . At very low temperatures nuclear spins were aligned with an applied magnetic field and the direction of emitted electrons were measured, as shown in Fig. 7.4. Notice that under a parity transformation, the velocity vector of the emitted electrons flips. However, the spin doesn’t flip – this is because spin is a type of vector called a pseudovector, which unlike regular vectors is invariant under a parity transformation. Wu found that beta emission occurred preferentially in the direction opposite to the nuclear spin, so parity conservation is violated.
Example 7.2.1 (Parity).
Question: A proton and anti-proton decay into two neutral pions via the interaction .
-
1.
Show the spherical harmonic (with , ) is an eigenfunction of parity and determine its eigenvalue.
-
2.
What is the parity of the initial state assuming the proton and anti-proton have zero orbital angular moment?
-
3.
The pions are produced with even orbital angular momentum. Determine the parity of the final state.
Solution.
1. In spherical coordinates
which is as . The eigenvalue is therefore +1.
2. These are fermions so . The parity is , giving .
3. These are bosons so . The parity is , giving .
Parity is violated so this cannot occur via the strong interaction.
7.2.3 Magnetic moment
From classical mechanics, the current caused by a charge moving with speed in a circle of radius includes a magnetic moment with magnitude
| (7.7) |
In quantum mechanics, we operationally define the observable magnetic moment to be in the direction of the greatest component of the angular momentum operator (which is with , leading to
| (7.8) |
where is the angular momentum quantum number. The Bohr and nuclear magnetrons are defined as
| (7.9) |
We can rewrite this as (for protons and neutrons)
| (7.10) |
where for protons and for neutrons (because neutrons have zero electric charge).
Since protons and neutrons have intrinsic (or spin) magnetic moments we can write
| (7.11) |
where (for protons and neutrons). Note is expected to be zero for the neutron (again since it has zero electric charge). For charged spin 1/2 point particles, such as the electron, . The quantity is called the spin g-factor.
7.3 The proton
It was soon realised by Antonius van den Broek that the positive nuclear charge (the atomic number ) corresponded to the position of the element in the periodic table. This was confirmed by Henry Moseley’s X-ray spectroscopy studies. In 1919 Rutherford suggested that the 1H nucleus is present in the nucleus of other elements. The basis for this was his observation of signatures of 1H in his scintillation detectors following bombardment of nitrogen gas with particles and his subsequent demonstration that this must have come from nitrogen nuclei. This marks the discovery of the proton.
Proton properties:
-
Mass: kg
-
Electric charge: +1e
-
Charge radius: 0.877 fm
-
Spin: 1/2
-
:
-
Parity: even
The proton is extremely stable with a half-life for decay of at least 6.6 y. Within the nucleus it can transform into a neutron via decay,
| (7.12) |
and electron capture,
| (7.13) |
However, a free proton will not decay on its own.
7.4 The neutron
It was known that the atomic mass number was generally about twice the atomic number . To account for this, the popular theory was that the nucleus contained protons, together with ‘nuclear’ electrons, leading to an overall charge of . Many arguments could be made against the existence of nuclear electrons and this ‘uncomfortable’ explanation for the disparity between nuclear charge and mass was resolved with the discovery of the neutron. Bombardment of beryllium with particles (generated by radioactive decay of radium) released a very penetrating but uncharged radiation, initially assumed to be rays. However, the energy of these rays was improbably high. It was Chadwick, working in Cambridge in 1932, who provided the correct interpretation:
| (7.14) |
Neutron properties:
-
Mass: kg
-
Electric charge: 0
-
Spin: 1/2
-
:
-
Parity: even
Unlike the proton, the free neutron is unstable with a t1/2 = 885.7 0.8 s:
| (7.15) |
This is the familiar decay process.
Neutrons can be generated by many nuclear reactions, for example, the reaction that was responsible for the discovery of the neutron (Eqn. 7.14). Small scale fission reactors are good sources of neutrons.
7.5 Anti-particles
The existence of anti-particles, usually designated by an overscore, is a general property of both fermions and bosons. The anti-particle has the same mass as the particle but opposite charge and magnetic moment. Fermions and anti-fermions can only be created (or destroyed) in pairs.
Thus in a proton collider, the following reaction is possible (if the beams are of sufficient energy to provide the rest mass of the newly created particles):
| (7.16) |
whereas the reaction:
| (7.17) |
is not. Perhaps the most familiar example of such a process is the pair production of an electron and a positron, the anti-particle of the electron, from a high energy ray, or the inverse process, the annihilation of a positron with an electron and the release of two 0.511 keV photons in opposite directions (to conserve momentum). The latter process is the physical basis of the PET scanner.
7.6 Exercises
Example 7.6.1.
If one interchanges the spatial coordinates of two electrons in a state (i) does the wave function change sign, or (ii) is the wave function unchanged?
Solution.
The parity of the two particle system is . Since and , the parity is even and so the wave function is unchanged under the exchange of spatial co-ordinates.
Example 7.6.2.
If one interchanges the spatial coordinates of an electron and positron in a state (i) does the wave function change sign, or (ii) is the wave function unchanged?
Solution.
The parity of the two particle system is . Since (an electron and positron have opposite parity since they are fermions) and , the parity is odd and so the wave function changes sign under the exchange of spatial co-ordinates.
